This is the legacy website for the Atomic and Molecular Data Unit.
The latest information about our activities can be found at
https://amdis.iaea.org.
Existing data services at this location function as usual.
About FLYCHK Rate coefficients
Methods
Direct Collisional Ionization
Excitation Autoionization
Radiative Recombination
Dielectronic Recombination
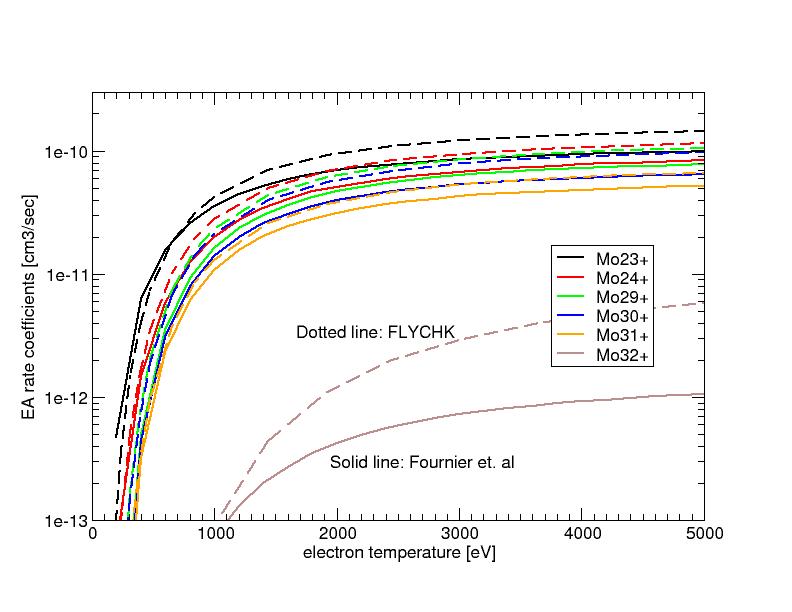
Comparison of Excitation Autoionization rate coefficients
Excitation Autoionziation rate coefficients of Molybdenum ions are compared with those of Fournier et al. [ Phys. Rev.A. 54 3870 (1996) ]. For most cases compared, FLYCHK values are in agreement with the published values within a factor of two or better except for Mo32+ ions where the difference is a factor of 5.
For this comparison, the two-electron collisional-excitation factor is set at 0.005. This factor may be too large for the L-shell ions such as Mo32+ (F-like ion). We plan to investigate this enhancement factor to reproduce Excitation Autoionization rate coefficients of more sophisticated calculations.
|
 |
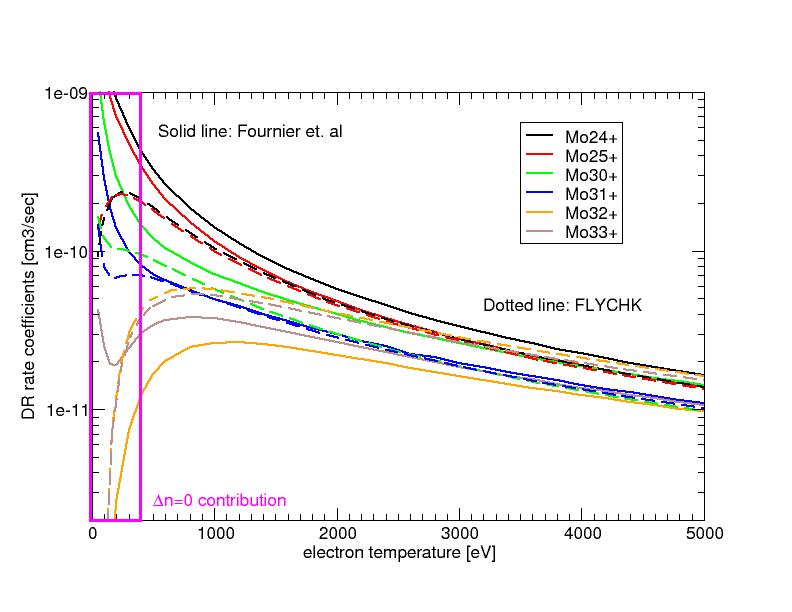
Comparison of Dielectronic Recombination rate coefficients
Dielectronic Recombination rate coefficients of Molybdenum ions are compared with those of Fournier et al. [Phys. Rev.A. 54 3870 (1996) ]. For most cases compared, FLYCHK values are in agreement witih the published values within a factor of two or better except for the low temperature cases where Δn=0 contributions of M-shell ions are dominant.
However, ions such as Mo24+ or Mo25+ ions have very low population fractions in those temperatures and hence the discrepancies of these ions at low temperatures will barely make a difference in charge state distributions. Neverthless, the effect of no Δn=0 contribution in FLYCHK calculations should be estimated and one must find a way to include in the code.
|
 |
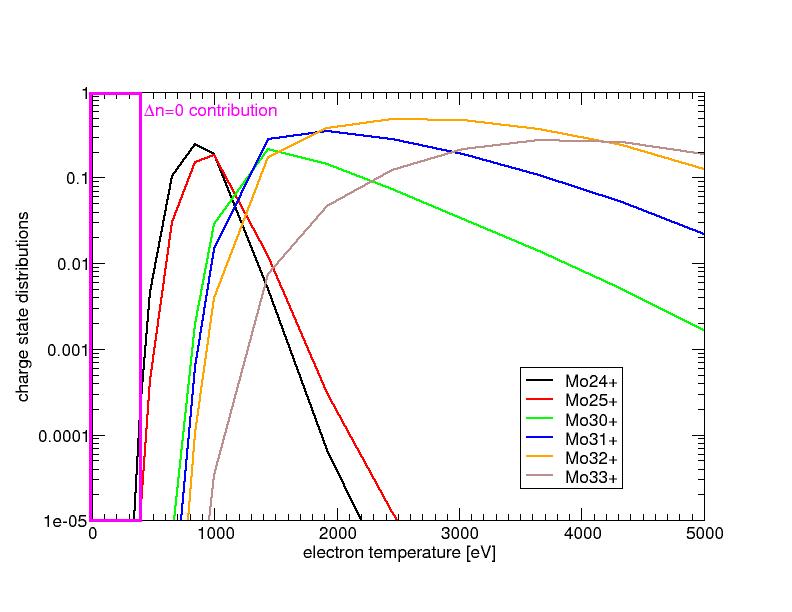
| Charge state distributions of Molebdenum ions are computed as a function of temperature in order to estimate the effect of uncertainties of Dielectron Recombination rate coefficients on the charge state distributions. The Mo24+ or Mo25+ ions have very low population fractions in low temperatures where the discrepancies between FLYCHK and Fournier results are the greatest. |
 |
References
1)
FLYCHK: an extension to the K-shell spectroscopy kinetics model FLY,
H. -K. Chung, W. L. Morgan and R. W. Lee, Journal of Quantitative Spectroscopy and Radiative Transfer, Volume 81, November 2003, Pages 107-115
2)
FLYCHK: Generalized population kinetics and spectral model for rapid spectroscopic analysis for all elements, H.-K. Chung, M.H. Chen, W.L. Morgan, Y. Ralchenko and R.W. Lee, High Energy Density Physics, Volume 1, Issue 1, December 2005, Pages 3-12